 
(credit a: modification of work by Tony Hisgett credit b: modification of work by “Atoma”/Wikimedia Commons) (a) One of the chemical properties of iron is that it rusts (b) one of the chemical properties of chromium is that it does not. Nitroglycerin is very dangerous because it explodes easily neon poses almost no hazard because it is very unreactive. Iron, for example, combines with oxygen in the presence of water to form rust chromium does not oxidize ( Figure 3). Examples of chemical properties include flammability, toxicity, acidity, reactivity (many types), and heat of combustion. The change of one type of matter into another type (or the inability to change) is a chemical property. (credit a: modification of work by “95jb14”/Wikimedia Commons credit b: modification of work by “mjneuby”/Flickr) (b) Steam condensing inside a cooking pot is a physical change, as water vapor is changed into liquid water. (a) Wax undergoes a physical change when solid wax is heated and forms liquid wax. In each of these examples, there is a change in the physical state, form, or properties of the substance, but no change in its chemical composition. Other examples of physical changes include magnetizing and demagnetizing metals (as is done with common antitheft security tags) and grinding solids into powders (which can sometimes yield noticeable changes in color). We observe a physical change when wax melts, when sugar dissolves in coffee, and when steam condenses into liquid water ( Figure 2). The different phase changes that matter can undergo. A physical change is a change in the state (Figure 1) or properties of matter without any accompanying change in its chemical composition (the identities of the substances contained in the matter), such as dissolution and dilution. Other physical properties, such as the melting temperature of iron or the freezing temperature of water, can only be observed as matter undergoes a physical change. We can observe some physical properties, such as density and color, without changing the physical state of the matter observed. Familiar examples of physical properties include density, color, hardness, melting and boiling points, and electrical conductivity. A physical property is a characteristic of matter that is not associated with a change in its chemical composition. 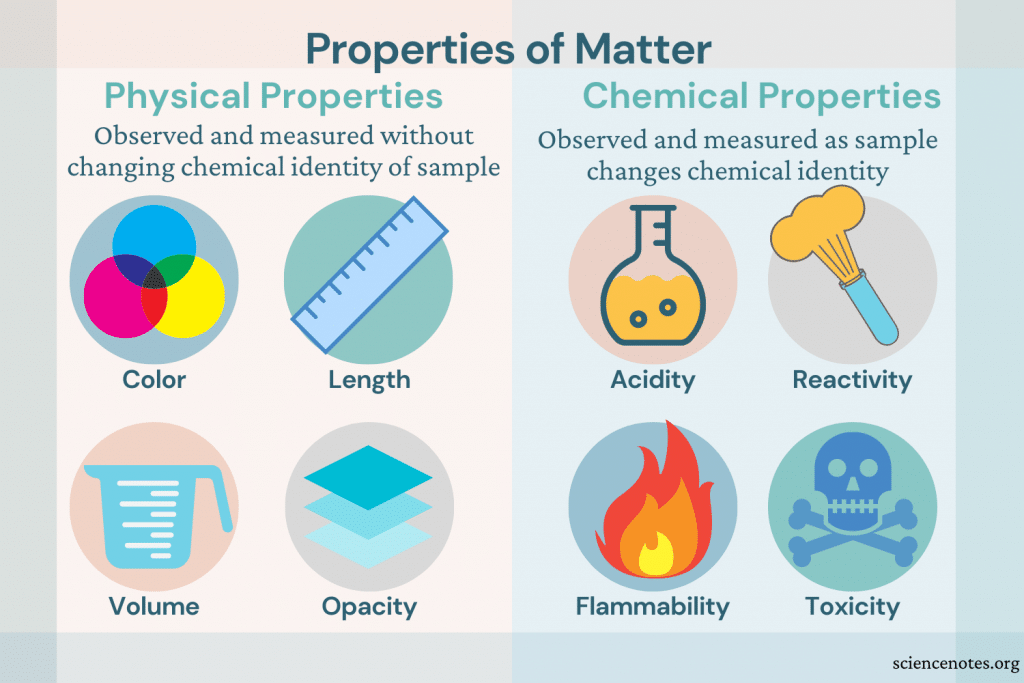
The characteristics that enable us to distinguish one substance from another are called properties. 
The faster they move, the “hotter” it is. If heat is needs to be supplied, the change is endothermic.Īn important distinction, is that heat is energy that flows due to a temperature difference, while temperature is a measure of the average kinetic energy of the molecules in a substance. When matter undergoes change, the process is often accompanied by a change in energy - heat, light, sound, kinetic energy of moving matter, etc… If heat is evolved during a change (is released) the change is exothermic. In this chapter, we’ll take a closer look at matter and energy and how they are related. Recall that chemistry is the study of matter, its properties, the changes that matter undergoes and the energy associated with these changes. Identify properties of matter as extensive or intensive.Identify properties of and changes in matter as physical or chemical.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
